News China
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

China Issues GB/T 46511, First National Standard for Digital Food Manufacturing Plants
China has released its first national standard for digital food manufacturing, marking a major step toward unifying technical guidance in the sector.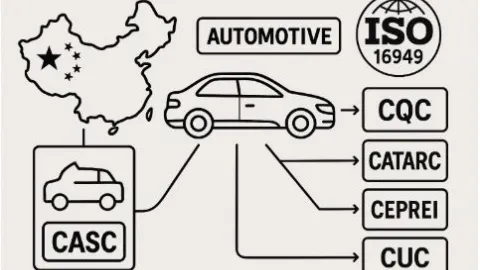
IATF Recognizes CQC Following Restructuring of Automotive Certification Bodies in China
The International Automotive Task Force (IATF) has reorganized its certification network in China by approving the separation of China Jiuding Automotive Supplier Certifications Co., Ltd. (CASC) into four independent certification bodies.
Hangzhou Summit Addresses Overseas Certification and Compliance for Chinese Automakers
The 2025 Global Automotive Regulations Certification Summit was held in Hangzhou on October 17–18, bringing together more than 500 participants from over ten countries.
China Quality Certification Center Among First Accredited for Personal Information Protection Compliance Audits
The China Quality Certification Center (CQC) has become one of the first professional institutions in China to receive accreditation for providing personal data protection compliance audits.
China Hosts World Standards Day Event, Showcases Standardization Initiatives
The 2025 World Standards Day main event was held in Beijing on October 14 under the theme “A Shared Vision for a Better World: Strengthening Partnerships for Sustainable Development.”
China Opens Consultation on New Rules for Online Catering Food Safety
The State Administration for Market Regulation (SAMR) has launched a public consultation on a draft regulation to strengthen food-safety responsibility for online catering platforms and the restaurants operating through them.
Nationwide Quality Month Campaign in China Continues with CQC Participation
The China Quality Certification Center (CQC) is continuing its nationwide activities for the 48th National Quality Month under the theme “Embracing Innovation and Green Growth, Safeguarding Quality.”
CNAS Prepares Random Supervision of Accredited Laboratories and Inspection Bodies for 2025
The China National Accreditation Service for Conformity Assessment (CNAS) will soon begin a round of random supervision for accredited laboratories and inspection bodies in 2025.
China Seeks Members for New ISO/IEC Directives Expert Group
The State Administration for Market Regulation (SAMR) has launched a call for experts to join a new group that will support China’s participation in the maintenance and revision of the ISO/IEC Directives.
Revised Chinese Forest Stewardship Standard Boosts Biodiversity and Climate Resilience
China will implement a major revision of its Forest Stewardship Standard (FSS) on January 1, 2026,
SAMR Seeks Proposals to Develop New ISO Standards for 2026
The Standard Innovation Department of the State Administration for Market Regulation (SAMR) in China is calling for
Quality Month Campaign in China Promotes Green Certification and Carbon Awareness
China Quality Certification Center (CQC) launched a series of activities in September as part of the 48th National Quality Month,Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,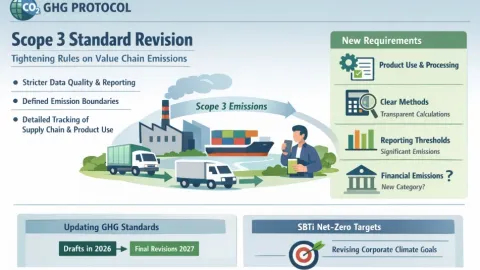
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.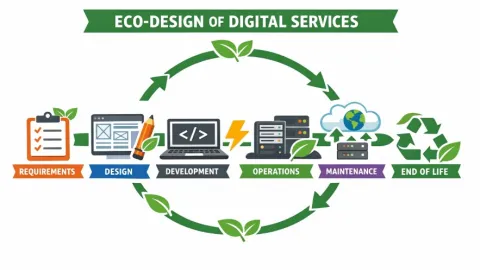
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,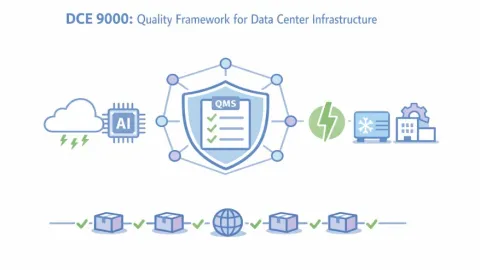
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

