News Netherlands
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Netherlands Takes the Reins on Global Carbon Capture, Transport, and Storage Standards
The Netherlands is driving international standardization of Carbon Capture, Utilisation and Storage (CCUS), with the Dutch Standardization Institute NEN coordinating efforts through the CEN/TC 474 committee.
New RvA Document Clarifies Rules for Flexible Accreditation Scopes in Netherlands
The Accreditation Council of the Netherlands (RvA) has published a new document, RvA-T054, that explains how flexible scopes of accreditation should be handled.
NEN Seeks Dutch Experts to Explore National Standards for Ayurveda and Yoga
The Netherlands is exploring whether there is enough interest to develop national standards for Ayurveda and yoga.
Zwolle Breaks Ground with First PEFC Trees outside Forests Certification
The municipality of Zwolle in the Netherlands has become the first to receive the Programme for the Endorsement of Forest Certification (PEFC) Trees outside Forests (TOF) certificate, marking a milestone in sustainable tree management beyond traditional forests.
NEN Seeks Dutch Experts for In Vitro Diagnostics Standards Committee
The Netherlands Standardization Institute (NEN) is seeking Dutch experts to join its In Vitro Diagnostics (IVD) standards committee.
NEN Initiates Development of Sustainability Guidelines for Medical Devices
In April 2025, the Netherlands Standardization Institute (NEN) will begin creating a Dutch Practical Guideline (NPR) focused on the sustainability of medical devices.
Dutch Experts Sought for New 'Chain of Custody' Standards
The Netherlands Standardization Institute (NEN) is seeking Dutch experts to contribute to the development of new 'chain of custody' standards, specifically ISO 13662 'Mass Balance' and ISO 13659 'Book and Claim'.
Dutch Involvement in European Sustainability Standards: Strong Foundation, But More Industry Support Essential
In a recent interview, Emiel Janssen, consultant at NEN, discussed the Netherlands’ role in shaping European sustainability standards.
Dutch Experts Sought for New European Circular Construction Standards
The European standards committee CEN/TC 350/SC 1 has established five new working groups to develop standards for circular design, material efficiency, and reuse in the construction sector.
Dutch Energy Sector Pushes for Circular Infrastructure as NEN Supports Transition
The Dutch energy sector is taking significant steps toward a circular economy to reduce the use of raw materials like aluminum, copper, and steel.
Dutch Initiative Aims to Close Standards Gap in Responsible Risk Profiling Algorithms
NEN, the Dutch standardization institute, is launching the development of a Dutch Technical Agreement (NTA) to guide organizations in the responsible use of risk profiling algorithms.
NEN Seeks Public Input on Revised Non-Standard Emission Measurement Guideline NPR 8117
The Netherlands Standardization Institute (NEN) is inviting stakeholders to provide feedback on the proposed revision of NPR 8117,Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,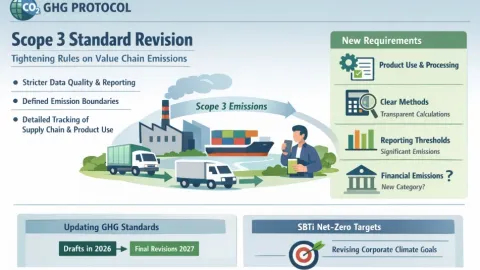
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

