News (Quality) Management
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Denmark Introduces Risk-Based Approach in Laboratory Accreditation Rules
DANAK, the Danish Accreditation Fund, has revised its AB 3 accreditation regulation for laboratories to align to relevant ILAC documents.
Three Quarters of Australian Labs Accredited to Latest Quality Requirements
More than 75% of laboratories in NATA’s accredited network have successfully transitioned to the ISO 15189:2022 standard, just 15 months after assessments began in August 2023.
Brazil Revises Requirements for Quality Management Systems
ABNT - Associação Brasileira de Normas Técnicas has revised its requirements for quality management systems to improve industry standards and align with evolving best practices.
Inmetro Advances in Quantum Measurement for Resistors, Calibration Fully Shifted to Brazil
Inmetro announced it has advanced its quantum methods for measuring standard resistors, making it possible to perform all calibrations in Brazil.
Spain’s Top Quality Organizations Meet to Boost Infrastructure Plans
The three most important bodies of Spain's standards system held a working meeting to advance the nation's infrastructure as part of the 2023 collaboration protocol.
Inmetro Accredits First Lab in Brazil's Northern Region
Inmetro has accredited the Laboratory of Product Testing (LabEP) at the University of the State of Amazonas (UEA).
Inmetro Strengthens Infrastructure Program in the North and Northeast Regions of Brazil
Inmetro is focusing its quality infrastructure program to support universities and institutes in Brazil’s North and Northeast regions.
KS CASCO Finalizes Serbian Translation of Standard for Guidelines for Workplace Safety Evaluation
The KS CASCO has approved the Serbian translation of ISO 45004:2024,
China Approves New Domestic Counterparts for ISO Technical Committees
China has officially approved new domestic organizations to serve as counterparts for five key ISO technical committees.
CAPS Australia’s ISO Certifications Prove Quality Isn’t Just Hot Air
"CAPS Australia", a leading provider of industrial air compressors and power generation equipment, has secured ISO 9001:2015 for quality management and ISO 45001:2018 for occupational health and safety.Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,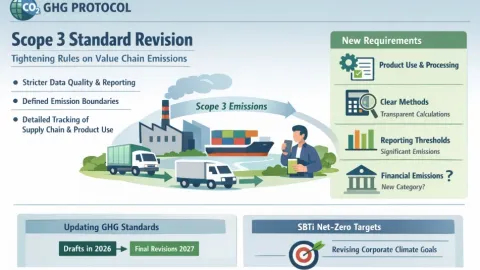
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

