News Slovenia
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Four Slovenian Companies Earn Green Construction Quality Label
Four companies in Slovenia have been awarded the Quality Label in Construction (ZKG) for their sustainable innovations in building products and technologies.
Kontron Earns Silver EcoVadis Award for Top-Tier Sustainability Performance
Kontron, d.o.o., a Slovenian telecommunications company, has been awarded the Silver Medal by EcoVadis, placing it among the top 15 percent of companies rated by the global sustainability assessment platform.
KOTO Reaffirms Commitment to Ethical and Sustainable Practices With SMETA Certification
KOTO, a Slovenian food processing firm, has once again passed the SMETA (Sedex Membership Ethical Trade Audit), one of the most widely used ethical audit formats globally.
"Zelena Slovenija" Organizes Free Online Conference on Sustainable Packaging and EU Rules
Zelena Slovenija is organizing a free professional conference on Innovations and Trends in Sustainable Packaging Solutions on March 27, 2025.
Slovenia Calls for Experts to Join Ongoing International and European Standardization Work
The Slovenian Institute for Standardization (SIST) is inviting experts to contribute to the development of new international and European standards across multiple industries.
Dreni Group SH.P.K, got PEFC certificate
Company from Priština, Kosovo, Dreni Group SH.P.K, got PEFC certificate as one of pioneer companies in Kosovo.
Prevekso Jeziki Achieves ISO 17100 Certification for Translation Service Quality
Prevekso Jeziki, a Slovenian language service provider and client of Bureau Veritas Slovenia, has obtained ISO 17100 certification, demonstrating its commitment to high-quality translation services.
March Exhibition by SIST to Highlight the Impact of Standards on Safety and Quality
The Slovenian Institute for Standardization (SIST) is hosting the official opening of the exhibition A Safe World – Embraced by Standards on March 26, 2025, at 17:00.
Slovenia Hosts Free Seminar on Standards for Importers and Businesses
The Slovenian Institute for Standardization (SIST) invites entrepreneurs to a free seminar on "The Importance of Standards for Businesses," scheduled for Thursday, March 20, 2025, at 10 a.m.
Topolšica Hospital Achieves Slovenia's First ISO 7101 Certification
Topolšica Hospital has become the first medical institution in Slovenia to earn the ISO 7101 certification for management systems for quality in healthcare organizations.
Deadline Extended for 33rd Quality and Excellence Conference in Portorož
The 33rd Conference on Quality and Excellence in Portorož has extended the deadline for paper submissions to January 24, 2025.
SIST Spreading the Team of Young Standardization Ambassadors
The Slovenian Institute for Standardization (SIST) has announced its new Standardization Ambassadors for 2025.Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,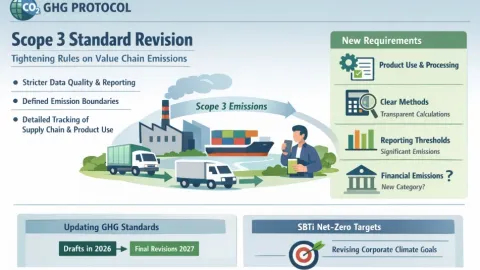
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

