News Czech Republic
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Czech National Awards Extend Application Deadline To April 21
The Ministry of Industry and Trade (MIT) and the Quality Council of the Czech Republic (RKČR) have extended the application deadline for the 26th National Awards of the Czech Republic from March 31 to April 21, 2026.
Quality Council Of The Czech Republic Publishes Newsletter Reviewing Quality Initiatives From 2025
The Quality Council of the Czech Republic and the Ministry of Industry and Trade (MPO) have published a newsletter summarizing the main activities carried out in 2025 to support quality, innovation, and responsible business practices in the country.
Czech Republic Proposes Technical Committee On Cybersecurity And Information Security
The Czech Standardization Agency (ČAS) has announced a proposal to establish a new technical standardization committee focused on cybersecurity and information security.
26th National Awards of the Czech Republic Invite Applications
The 26th National Awards of the Czech Republic have officially opened, inviting organizations to apply for one of the country’s established national recognition programs focused on quality, responsibility, and sustainability.
National Program Czech Quality Newsletter Reviews 2025 Activities and New Quality Labels
The National Program Czech Quality, a government-backed initiative of the Czech Republic,
ČSNMap Portal Goes Live, Linking Technical Standards With Czech Building Law
The Czech Standardization Agency (ČAS) has launched the ČSNMap portal, a digital tool that for the first time directly links Czech Technical Standards (ČSN) with Czech building legislation.
Fifth Quality Conference in the Czech Republic Connects Nuclear Energy, Healthcare And Automotive Sectors
The fifth edition of the Quality Conference was held on November 25–26, 2025, at Hotel Zámek Valeč in Valeč, in the Karlovy Vary Region of the Czech Republic,
GORDIC GINIS Filing Service Earns First eSSL Certification in Czechia
The GINIS electronic filing service from GORDIC has become the first system in Czechia to earn eSSL certification.
25th National Awards of the Czech Republic Honor ČEZ and Prague 13 as Absolute Winners
ČEZ and the Prague 13 Municipal District were named the absolute winners of the 25th National Quality Awards of the Czech Republic at a formal award ceremony held on November 20, 2025, in the Main Hall of the Wallenstein Palace in Prague.
You Can’t Replace Czech Quality: November Episode Highlights Trust in Quality Brands
The November episode of You Can’t Replace Czech Quality series explains why Czech quality remains a trusted sign of reliable products and services.
Prague to Host 25th National Quality Awards of the Czech Republic
The 25th edition of the Czech Republic’s National Quality Awards will take place on November 20, 2025, in the Main Hall of the Wallenstein Palace, seat of the Senate of the Parliament of the Czech Republic.
Fifteen Czech Companies Introduced to the National Quality Program
Fifteen Czech companies were introduced this year to the government-backed National Program Česká kvalita during the Day with Quality event at the Ministry of Industry and Trade (MPO).Global News

AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).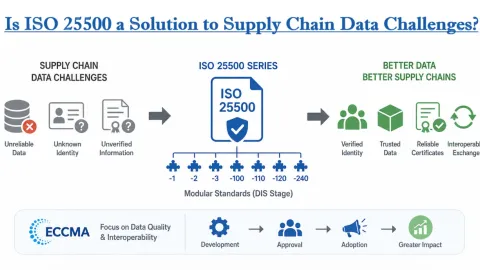
Is ISO 25500 a Solution to Supply Chain Data Challenges?
ISO 25500, a draft series of standards from the International Organization for Standardization (ISO), is presented by the Electronic Commerce Code Management Association (ECCMA) as a possible way to address long-standing problems with unreliable data across global supply chains.
IFS Updates Food, Logistics, Broker And Pacsecure Doctrines To Clarify Certification Rules
The International Featured Standards (IFS) has published updated doctrine documents for its current standards covering food production, logistics, brokerage, and packaging materials, clarifying how certification requirements should be applied without revising the standards themselves.
PAS 25535 on Sustainability in Asset Management Reaches Draft Stage
PAS 25535, a specification focused on integrating sustainability into asset management systems, has reached the committee draft stage within the International Organization for Standardization (ISO), marking the close of its initial comment period.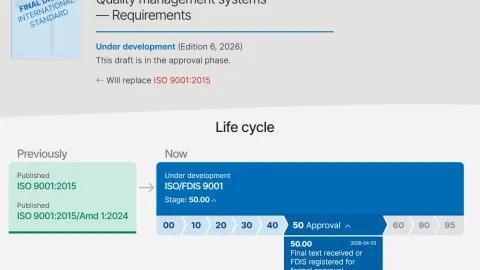
ISO 9001 Revision Moves to FDIS Stage as Early Analysis Questions Scope of Changes
The International Organization for Standardization (ISO) has confirmed that the revision of ISO 9001 has entered the Final Draft International Standard (FDIS) stage, marking the last step before publication.
DIN to Lead ISO IEC Joint Technical Committee 5 on Digital Product Passport Standards
Following an initiative by the German Institute for Standardization (DIN) and the German Commission for Electrical, Electronic & Information Technologies (DKE),
ANSI/TIA-942 Addendum Targets AI Data Center Cooling and High-Density Needs
The Telecommunications Industry Association (TIA) is developing a new addendum to the ANSI/TIA-942 data center standard to address the growing demands of artificial intelligence (AI) infrastructure.

