News South America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

IRAM Presents New Conformity Assessment Seals
The Argentine Institute of Standardization and Certification (IRAM) has renewed its conformity assessment seals after more than 60 years.
ABNT Launches Standard for Open Data in Smart Cities at São Paulo Event
The Associação Brasileira de Normas Técnicas (ABNT) has officially released ABNT NBR ISO 37110:2024,
Brazil Confirms Quality Strategy Implementation with Official Resolutions
Brazil has formally enacted its National Quality Infrastructure Strategy (ENIQ) and the 2025–2026 Action Plan through Resolutions No. 1 and No. 2 of 2025, published in the Official Gazette on July 1.
Brazil Opens Consultation to Update Product Certification Requirements
Brazil’s National Institute of Metrology, Quality and Technology (Inmetro) has launched a public consultation to revise the country’s general rules for product certification.
Inmetro Puts Digital Verification in the Palm of Your Hand
The Brazilian government has officially launched Inmetro na Palma da Mão (“Inmetro in the Palm of Your Hand”), a new digital verification platform that allows consumers to confirm whether certified products are genuine.
Brazil Unveils First National Quality Strategy Action Plan for 2025–2026
Brazil has launched its National Quality Infrastructure Strategy (ENIQ) and the first related Action Plan, which will be in effect for the 2025–2026 period.
Nominations Now Open for the BRCGS Awards 2025 in the Americas Region
Nominations are now open for the BRCGS Awards 2025, which will be presented at BRCGS Connect Charlotte 2025 this September in North Carolina.
Inmetro Earns Top Fiesp Regulatory Quality Seal from São Paulo Industry Federation
The National Institute of Metrology, Quality, and Technology (Inmetro) has received the 2024 Gold Category of the Regulatory Quality Seal from the Federation of Industries of the State of São Paulo (Fiesp)—
IRAM Marks 90 Years of Advancing Quality and Safety in Argentina
The Argentine Institute of Standardization and Certification (IRAM) celebrated its 90th anniversary on May 2, highlighting nearly a century of work supporting quality, safety, and sustainability in Argentina and beyond.
Federal Prosecutors in Brazil Recognized with Top Certification for Gender Violence Prevention
The Brazilian Association of Technical Standards (ABNT) has awarded the National Council of the Public Prosecutor’s Office (CNMP) the highest category of the “Nós Por Elas” Seal, recognizing its efforts to prevent and combat violence against women.
Public Labs Across Brazil Can Now Join Inmetro’s ProdIQ Accreditation Program
Public institutions throughout Brazil can now voluntarily join the ProdIQ Program, which promotes the accreditation of testing and calibration laboratories and supports professional training.
Lagoa Dourada Recognized as Sustainable City by ABNT
Lagoa Dourada, a municipality of about 12,000 people in Minas Gerais, has been officially recognized as a Sustainable City by the Brazilian Association of Technical Standards (ABNT).Global News

ISO 22000 Revision Enters Public Enquiry Stage
The International Organization for Standardization (ISO) has moved the revision of ISO 22000 to the public enquiry stage.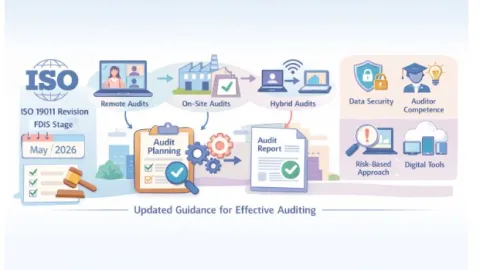
ISO 19011 Auditing Standard Enters Final Stage With Hybrid And Digital Shift
The International Organization for Standardization (ISO) has advanced the revision of ISO 19011 to the Final Draft International Standard (FDIS) stage.
IMDRF Playbook Guides Global Use Of Shared Medical Device Regulatory Reviews
The International Medical Device Regulators Forum (IMDRF) has published a playbook to help regulators use shared approaches when reviewing medical devices, known as regulatory reliance.
DIN Introduces First Framework For AI Testing Tools
The German Institute for Standardization (DIN) has published DIN SPEC 92006, introducing the first unified framework for artificial intelligence (AI) testing tools and setting clear requirements for how these tools should be designed and used.
FSSC 22000 Version 7 Release Moved To May 2026
The Foundation for Food Safety Certification (FSSC) has delayed the launch of FSSC 22000 Version 7, with publication now expected in early May 2026.
FSC Launches Normative Newsfeed For Clearer Requirements Updates
The Forest Stewardship Council (FSC) has introduced a new Normative Newsfeed to make updates to its requirements easier to track and understand.
OECD Report Links Standards To More Effective Regulation
The Organisation for Economic Co-operation and Development (OECD) has released a report examining how governments can improve regulation by making better use of standards, measurement, and assurance systems, known as quality infrastructure (QI).
ISO 9000 Revision Reaches Final Draft Stage, Publication Expected In May 2026
The revision of ISO 9000, the international standard that defines the basic concepts and vocabulary of quality management, has reached the Final Draft International Standard (FDIS) stage, the final step before publication.
ISO and IEC Form JTC 5 Committee on Digital Product Passport Standards
A new international standards committee has been created to develop standards for digital product passports (DPPs), systems designed to share key product information across the entire lifecycle and improve transparency and circular economy practices.
Third Edition of ISO 22301 Business Continuity Standard Enters Development
The next revision of ISO 22301, the international standard for business continuity management systems (BCMS), has moved into development.
Global Accreditation Cooperation Incorporated Launches Website
Global Accreditation Cooperation Incorporated has launched its new official website, providing a central online platform for the organization that became the single global body for international accreditation cooperation.

