News United States of America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Medical Device Industry at Midpoint in Transition to FDA’s ISO 13485-Based QMSR
A year ago, the FDA adopted ISO 13485 into its Quality System Regulation, rebranding it as the Quality Management System Regulation (QMSR).
"Beyond Celiac" Unveils New Trademark for Gluten-Free Products
Beyond Celiac, a leading gluten-free association in the United States, has introduced a new trademark following the release of Issue 4 of the BRCGS Standard.
ANAB Seeks Experts for Personnel Certification and Certificate Accreditation Committees
The ANSI National Accreditation Board (ANAB) is inviting nominations for its Personnel Certification Accreditation Committee (PCAC) and Certificate Accreditation Program Accreditation Committee (CAPAC).
NIST Boosts Textile Recycling Efforts with Comprehensive NIR-SORT Database
The National Institute of Standards and Technology (NIST) has developed the Near-Infrared Spectra of Origin-defined and Real-world Textiles (NIR-SORT), a free and downloadable database designed to improve textile sorting efficiency at recycling centers.
ANAB Announces Inaugural Conformity Assessment Symposium for 2026
The ANSI National Accreditation Board (ANAB) will host its first Conformity Assessment Symposium (ACAS) on January 19-20, 2026, at the Gaylord Palms Resort & Convention Center in Orlando, Florida.
Eurofins Introduces GMP Certification Program for Dietary Supplements
Eurofins Healthcare Assurance has launched a new Good Manufacturing Practice (GMP) certification program to help dietary and food supplement companies meet U.S. regulatory standards.
SCC and A2LA Launch First Dual Accreditation Program for North America
The Standards Council of Canada (SCC) and the American Association for Laboratory Accreditation (A2LA) have introduced a dual accreditation program to help testing and calibration laboratories achieve seamless cross-border compliance.
Private Brands Increase Market Share, Approach Quarter of Retail Sales
Private brands now account for nearly a quarter- 24% - of retail sales in ten main product sectors, according to recent research by Numerator.
FDA Overhauls ‘Healthy’ Food Labeling with Stricter Criteria
The U.S. Food and Drug Administration (FDA) has issued a final rule updating the criteria for the “healthy” nutrient content claim on food packaging.
ANSI Invited Experts to Shape the Future of Sustainability Standards
The American National Standards Institute (ANSI) called for experts on January 3, 2025, to join a Virtual Technical Advisory Group (VTAG) and contribute to the revision of ISO Guide 82:2019,
Applications Open for 2025 Baldrige Award, the Highest U.S. Honor in Organizational Excellence
The National Institute of Standards and Technology (NIST) has announced the 2025 Baldrige Award Criteria, signaling the start of the application process for the nation’s highest recognition for organizational performance.
Mariposa Labs Earns ISO 22716 Certification for Cosmetic Manufacturing
Mariposa Labs, a personal care contract manufacturer based in Boise, Idaho, has achieved ISO 22716:2007 certification.Global News

ISO 45001 Second Committee Draft Version Highlights Safety Culture and Workplace Wellbeing
The revision of ISO 45001 has advanced with the publication of the second version of the first Committee Draft, maintaining the standard at the Committee Draft stage and reflecting feedback from the first international enquiry.
ISO 23662 on Vegetarian and Vegan Foods Enters Periodic Review
ISO 23662:2021, the international standard that defines criteria for vegetarian and vegan foods, has entered its periodic review phase, giving stakeholders the opportunity to decide whether it should remain unchanged or be revised.
PEFC Circular Materials Task Force Established, Experts and Stakeholders Invited to Apply
The Programme for the Endorsement of Forest Certification (PEFC) has established its PEFC Circular Materials Task Force and is inviting nominations for membership by March 18, 2026.
ASTM Develops WK91420 Guide for Artificial Intelligence in HR
ASTM International is developing a new guide, WK91420, to set clearer standards for artificial intelligence (AI) in recruitment and hiring, aimed at reducing bias and improving fairness.
ISO 9001 Revision Advances After Review of 1,700 DIS Comments
The revision of ISO 9001 has moved into a decisive stage, with international experts completing the review of comments on the Draft International Standard (DIS) and confirming that the new edition is on track for publication in September 2026.
ISO Launches ISO 14019 Series With New Standards Supporting Validation and Verification of Sustainability Information
The International Organization for Standardization (ISO) has published the first three standards launching the ISO 14019 series,
ISO 18060 Sets Global Rules to Measure Tourism Environmental and Social Impact
ISO 18060, a newly published international standard from January 2026, sets out a global framework to help tourism organizations measure their environmental, social, human, and economic impacts in a consistent and comparable way.
IFS Introduces Local Check to Support Small-Scale Food Producers
International Featured Standards (IFS) has introduced IFS Local, a new annual standalone food safety check designed to help small-scale and artisanal food producers demonstrate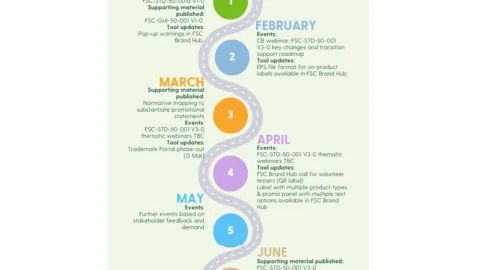
FSC Releases Revised Trademark Standard and Starts Rollout for Certificate Holders
The Forest Stewardship Council (FSC) has released a revised trademark standard and has started rolling out guidance and support materials to help organizations transition to the updated requirements.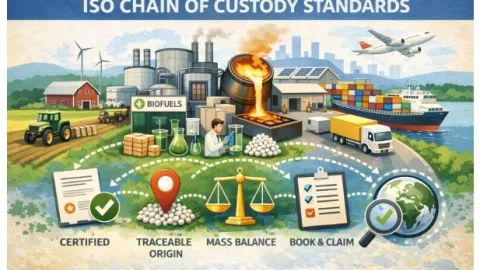
ISO 22095 Parts 2 And 3 Define Rules For Material Tracking Chain Of Custody Models
The International Organization for Standardization (ISO) has published ISO 22095-2 and ISO 22095-3, two new international standards that
CSR Labels and ISO 26000 Linked to Improved Business Performance
A new international study by Responsibility Europe finds that organizations using recognized CSR (corporate social responsibility) labels outperform others economically and organizationally, confirming the growing importance of CSR frameworks and ISO 26000 worldwide.

