News United States of America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Spring 2026 ‘ANSI in China’ Newsletter Highlights Standards and Policy Developments
The American National Standards Institute (ANSI) has published the Spring 2026 edition of its ANSI in China Newsletter in English and Chinese.
ANSI Starts National Effort To Align Critical Minerals Standards Across The Supply Chain
The American National Standards Institute (ANSI) has launched a multi-phase initiative to coordinate standards for critical minerals, aiming to strengthen U.S. supply chains and reduce reliance on foreign sources.
Quantum Standards Lag Behind Technology Growth, ASCET Scan Finds
The Advancing Standardization for Critical and Emerging Technologies (ASCET) Center of Excellence (CoE) has found in a new environmental scan that quantum standardization is not keeping pace with rapid technological progress,
ANAB Launches QAEP to Evaluate Credibility and Effectiveness of Quality Assurance Schemes
The ANSI National Accreditation Board (ANAB) has launched the Quality Assurance Evaluation Program (QAEP),
Closing The Gap In Process Safety Controls Outside SIS With New ISA Framework
The International Society of Automation (ISA) has introduced ANSI/ISA-84.91.03,
ANSI Outlines Implementation Plan for U.S. Standards Strategy 2025
The American National Standards Institute (ANSI) has outlined how it will move the U.S. Standards Strategy 2025 (USSS) into implementation,
ASCET Report Flags Gaps In Quantum Standards, Definitions And Benchmarking
Gaps in definitions and performance benchmarks are slowing the development and adoption of quantum technologies,
USDA Campaign Highlights ‘Product of USA’ Label With Full-Origin Requirement
The U.S. Department of Agriculture (USDA) has begun a national campaign to promote its voluntary “Product of USA” label, now limited to meat, poultry, and egg products fully produced in the United States.
FDA Schedules Town Hall On Risk Based Inspections Under New QMSR Framework
The U.S. Food and Drug Administration (FDA) has announced a Town Hall on April 1, 2026, to explain how inspections of medical device manufacturers will change under its new Quality Management System Regulation (QMSR), with a focus on a risk-based approach.
FDA Shifts Medical Device Inspections To Lifecycle-Based QMS Oversight
The U.S. Food and Drug Administration (FDA) has updated how it inspects medical device manufacturers, introducing a lifecycle‑based approach to quality management system oversight under its new Quality Management System Regulation.
ANSI Invites Experts To Lead U.S. TAG For Automation Standards
The American National Standards Institute (ANSI) is inviting experienced automation professionals to consider leadership roles in the U.S. Technical Advisory Group (TAG) to ISO/TC 184/SC 4, which contributes to international standards for automation systems and industrial data.
NIST Launches AI Agent Standards Initiative To Address Security And Interoperability
The National Institute of Standards and Technology (NIST) has launched an AI Agent Standards Initiative to address security, identity, and interoperability challenges as AI agents become more widely used.Global News

FSSC 22000 Version 7 Updates Prerequisite Programs And Aligns With GFSI 2024
The Foundation FSSC has released Version 7 of the FSSC 22000 food safety management system certification scheme, introducing updated prerequisite programs (PRPs) and aligning the scheme with the 2024 Global Food Safety Initiative requirements.
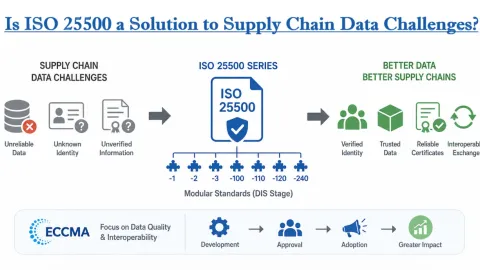
ISO Develops Carbon Credit Data Model Standard, Calls for Input
A new data model for carbon credit markets is being developed by ISO’s sustainable finance and financial services committees, with early input still open to stakeholders.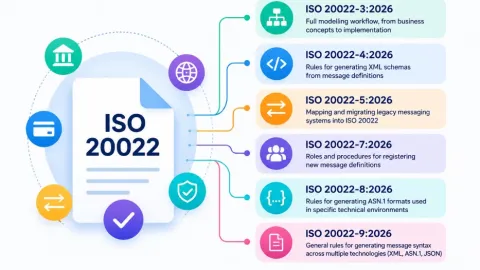
ISO 20022 Series Update Sets New Rules as Payment Systems Prepare for Implementation
The International Organization for Standardization (ISO) has published updated and new parts in the ISO 20022 series, as major payment systems advance their implementation of the standard on closely aligned timelines.
Consultation Underway On Global Standard For AI Testing In Medical Devices
An international voting and consultation phase is underway for a new standard that sets requirements for testing artificial intelligence (AI) and machine learning (ML) in medical devices.
MSC Opens Review Of Chain Of Custody Standard For Public Consultation
The Marine Stewardship Council (MSC) has launched a public consultation on proposed revisions to its Chain of Custody Standard, inviting stakeholders across the seafood supply chain to provide feedback until May 30, 2026.
Call For Food Sector Stakeholders To Influence ISO 22000 Revision Through National Committees
Food sector stakeholders who apply ISO 22000 in practice have the opportunity to influence its revision by bringing real-world experience into the process.
AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).