News Balkan
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Serbia Opens Public Review of Serbian Translation of Updated ISO 14001
The Institute for Standardization of Serbia (ISS) has opened a public consultation on the draft naSRPS EN ISO 14001:2026, inviting experts to comment on the Serbian translation of the updated environmental management standard.
Montenegro Adopts 2026 Plan for National Standards Development
The Institute for Standardization of Montenegro (ISME) has adopted its annual plan for 2026, setting out the standards and related documents to be introduced, updated, or reviewed during the year.
Romania Adopts ISO 14001:2026 Environmental Management Standard
The Romanian Standards Association (ASRO) has published SR EN ISO 14001:2026, bringing the latest version of the environmental management systems standard into national use.
Serbia Updates Accreditation Requirements for Organic Product Certification Bodies
The Accreditation Body of Serbia (ATS) has announced updates to accreditation scopes for certification bodies in organic food production following the adoption of a new Law on Organic Production, which takes effect on June 1, 2026.
ISBIH Hosts Webinar on New Version of ISO 14001
The Institute for Standardization of Bosnia and Herzegovina (ISBIH) will host a free webinar on April 15, 2026, to present the upcoming changes in ISO 14001, the environmental management systems standard.
ICT, Telecommunications And Education in New ISS Standardization Bulletin
The Institute for Standardization of Serbia (ISS) has released a new issue of its Standardization bulletin, highlighting how standards support digital infrastructure, education, and management practices.
Croatian Standards Institute Hosts 5th Expert Council Session in Zagreb
The Croatian Standards Institute (HZN) held the 5th session of its Expert Council in Zagreb, bringing together 269 members to strengthen cooperation and participation in standardization.
Greece Opens Public Consultation on 58 Revised Technical Specifications for Public Works
The Hellenic Organization for Standardization (ELOT) has launched a public consultation on 58 revised Greek Technical Specifications that will become the updated, second edition of national requirements for public works.
Serbia Hosts 35th Quality Week Spotlighting Green Transition AI and Risk Management
The 35th Quality Week in Serbia brought together institutions, experts, and industry representatives to explore topics such as green transition, artificial intelligence in healthcare, and risk management in modern organizations.
Montenegro Accreditation Body Completes EA MLA Follow-Up Evaluation
The Accreditation Body of Montenegro (ATCG) underwent a follow-up evaluation in March 2026 as part of the European co-operation for Accreditation (EA) Multilateral Agreement (MLA) process.
Academic Standards Day Connects Students With Standards And Career Opportunities In Belgrade
The Institute for Standardization of Serbia (ISS) has announced that Academic Standards Day will take place in Belgrade on April 1,
Montenegro Launches National Technical Committee for Building Information Modeling in Construction
The Institute for Standardization of Montenegro (ISME) has launched the formation of a national technical committee dedicated to building information modeling (BIM),Global News

FSSC 22000 Version 7 Updates Prerequisite Programs And Aligns With GFSI 2024
The Foundation FSSC has released Version 7 of the FSSC 22000 food safety management system certification scheme, introducing updated prerequisite programs (PRPs) and aligning the scheme with the 2024 Global Food Safety Initiative requirements.
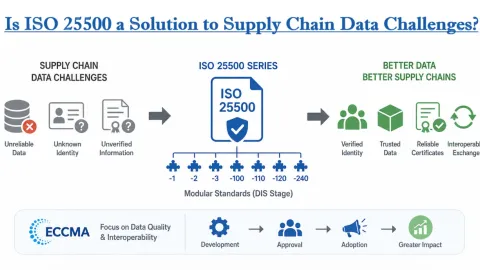
ISO Develops Carbon Credit Data Model Standard, Calls for Input
A new data model for carbon credit markets is being developed by ISO’s sustainable finance and financial services committees, with early input still open to stakeholders.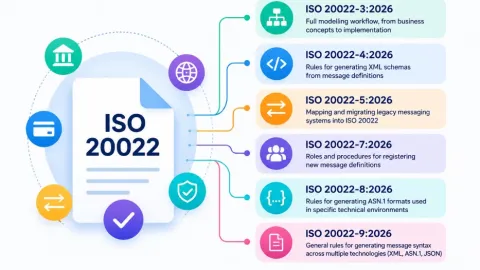
ISO 20022 Series Update Sets New Rules as Payment Systems Prepare for Implementation
The International Organization for Standardization (ISO) has published updated and new parts in the ISO 20022 series, as major payment systems advance their implementation of the standard on closely aligned timelines.
Consultation Underway On Global Standard For AI Testing In Medical Devices
An international voting and consultation phase is underway for a new standard that sets requirements for testing artificial intelligence (AI) and machine learning (ML) in medical devices.
MSC Opens Review Of Chain Of Custody Standard For Public Consultation
The Marine Stewardship Council (MSC) has launched a public consultation on proposed revisions to its Chain of Custody Standard, inviting stakeholders across the seafood supply chain to provide feedback until May 30, 2026.
Call For Food Sector Stakeholders To Influence ISO 22000 Revision Through National Committees
Food sector stakeholders who apply ISO 22000 in practice have the opportunity to influence its revision by bringing real-world experience into the process.
AutoCarbon Certification Scheme Due in 2027 As Pilot Phase Begins
The International Automotive Oversight Bureau (IAOB) has announced AutoCarbon,
ISO 14064-5 Introduces Guidance For Remote Verification Of Greenhouse Gas Data
The International Organization for Standardization (ISO) has introduced ISO 14064-5, which provides guidance on how to carry out verification and validation of greenhouse gas (GHG) data using remote techniques in digital environments.
ISO 9002 Revision Moves To Draft International Standard Stage
The revision of ISO 9002 has entered the Draft International Standard (DIS) stage, with ISO/TC 176/SC 2, the ISO subcommittee responsible for quality systems, advancing the project into formal consultation.
ISO 37304 Compliance Management Certification Standard Advances to Final Draft Stage
The International Organization for Standardization (ISO) has advanced ISO 37304 to the final draft international standard (FDIS) stage.
One Vote Tips Toward New Edition Of ISO/IEC 17065 Over Amendment
One vote has tipped the decision toward publishing a new edition of ISO/IEC 17065 instead of issuing a separate amendment, following a closely split ballot within the International Organization for Standardization (ISO).