News Europe
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

Slovenia Introduces Flag Label On Food To Guarantee Full Domestic Origin
The Ministry of Agriculture, Forestry and Food of Slovenia (MAFF) has introduced a new label featuring the national flag to guarantee that food products are entirely produced, processed, and packaged in Slovenia.
EUDOM Achieves ISO 14001 Certification For Environmental Management
EUDOM, d.o.o., part of the EUTRIP group, has obtained certification to ISO 14001, the international standard for environmental management systems.
Croatia Calls On SMEs To Join EU Standardization Regulation Workshop
The Croatian Standards Institute (HZN) is encouraging small and medium-sized enterprises (SMEs) to take part in a European Commission (EC) workshop on the revision of the EU Standardization Regulation (EU) No 1025/2012.
Italy Opens UNI 12000 Certification Scheme for Anti-Bullying Systems
Accredia, Italy’s national accreditation body, has opened accreditation for certification bodies that will certify organizations against UNI 12000,
Free Risk Tool Launched for Healthcare Information Security Standard NEN 7510
The Netherlands Standardization Institute (NEN) has launched a free risk analysis tool to help healthcare organizations and other entities that process personal health data carry out the information security risk assessment required under NEN 7510.
AI System Testing Guidance DIN SPEC 92006 Featured In Latest DIN Newsletter
The German Institute for Standardization (DIN) has released a new issue of its DINews newsletter, highlighting DIN SPEC 92006, a specification designed to help organizations test and evaluate artificial intelligence (AI) systems.
TÜV AUSTRIA Certified To ISO 30415 As Austria Prepares For EU Pay Transparency Rules
TÜV AUSTRIA, an Austrian testing, inspection, and certification organization, has obtained certification to ISO 30415,
Kvalitetsmagasinet Opens Voting For Sweden’s 2026 Quality Profile Award
Kvalitetsmagasinet, a Swedish publication focused on quality management, has opened voting for the 2026 edition of its annual “Quality Profile of the Year” award after receiving 64 nominations from across the country.
SKZ Warns About Fake Certificates For Plastic Products
SKZ - German Plastics Center has warned about fake SKZ certificates for plastic products appearing in the market.
UNE Featured In CEOE Environmental Practices Catalog For Circular Economy Initiative
The Spanish Association for Standardization (UNE) has been featured in the 2025 Environmental Best Practices Catalog of Spanish Companies,
Quality Council Of The Czech Republic Publishes Newsletter Reviewing Quality Initiatives From 2025
The Quality Council of the Czech Republic and the Ministry of Industry and Trade (MPO) have published a newsletter summarizing the main activities carried out in 2025 to support quality, innovation, and responsible business practices in the country.
ISO 21800: Making Sense Of Long And Confusing Online Terms And Conditions
Long and complex online terms and conditions often discourage consumers from reading them, creating a gap between what people agree to and what they actually understand.Global News

ISO 45001 Second Committee Draft Version Highlights Safety Culture and Workplace Wellbeing
The revision of ISO 45001 has advanced with the publication of the second version of the first Committee Draft, maintaining the standard at the Committee Draft stage and reflecting feedback from the first international enquiry.
ISO 23662 on Vegetarian and Vegan Foods Enters Periodic Review
ISO 23662:2021, the international standard that defines criteria for vegetarian and vegan foods, has entered its periodic review phase, giving stakeholders the opportunity to decide whether it should remain unchanged or be revised.
PEFC Circular Materials Task Force Established, Experts and Stakeholders Invited to Apply
The Programme for the Endorsement of Forest Certification (PEFC) has established its PEFC Circular Materials Task Force and is inviting nominations for membership by March 18, 2026.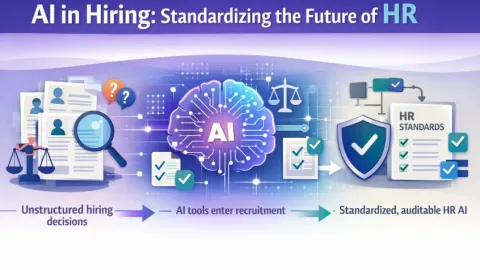
ASTM Develops WK91420 Guide for Artificial Intelligence in HR
ASTM International is developing a new guide, WK91420, to set clearer standards for artificial intelligence (AI) in recruitment and hiring, aimed at reducing bias and improving fairness.
ISO 9001 Revision Advances After Review of 1,700 DIS Comments
The revision of ISO 9001 has moved into a decisive stage, with international experts completing the review of comments on the Draft International Standard (DIS) and confirming that the new edition is on track for publication in September 2026.
ISO Launches ISO 14019 Series With New Standards Supporting Validation and Verification of Sustainability Information
The International Organization for Standardization (ISO) has published the first three standards launching the ISO 14019 series,
ISO 18060 Sets Global Rules to Measure Tourism Environmental and Social Impact
ISO 18060, a newly published international standard from January 2026, sets out a global framework to help tourism organizations measure their environmental, social, human, and economic impacts in a consistent and comparable way.
IFS Introduces Local Check to Support Small-Scale Food Producers
International Featured Standards (IFS) has introduced IFS Local, a new annual standalone food safety check designed to help small-scale and artisanal food producers demonstrate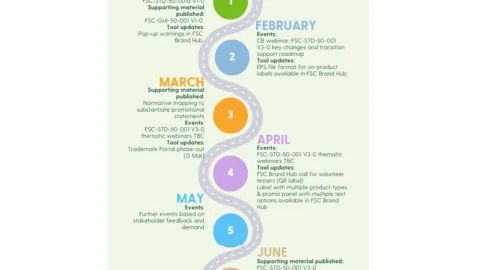
FSC Releases Revised Trademark Standard and Starts Rollout for Certificate Holders
The Forest Stewardship Council (FSC) has released a revised trademark standard and has started rolling out guidance and support materials to help organizations transition to the updated requirements.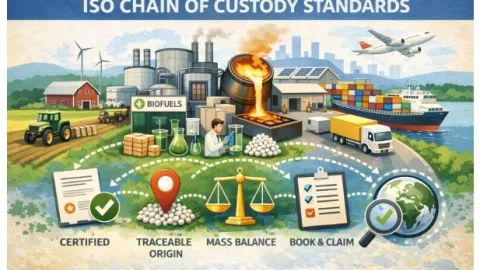
ISO 22095 Parts 2 And 3 Define Rules For Material Tracking Chain Of Custody Models
The International Organization for Standardization (ISO) has published ISO 22095-2 and ISO 22095-3, two new international standards that
CSR Labels and ISO 26000 Linked to Improved Business Performance
A new international study by Responsibility Europe finds that organizations using recognized CSR (corporate social responsibility) labels outperform others economically and organizationally, confirming the growing importance of CSR frameworks and ISO 26000 worldwide.

