News United States of America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

FDA Invites Comment on Third-Party Food Safety Certification Rule
The U.S. Food and Drug Administration (FDA), the federal agency responsible for overseeing food safety and regulating imports,
Students Challenged to Explore Hidden Role of Standards in 15th ANSI Paper Competition
The American National Standards Institute (ANSI) and its Committee on Education have launched the 15th annual Student Paper Competition,
NESC 2028 Revision Cycle Moves Into Public Comment Phase
The National Electrical Safety Code (NESC), the United States’ framework for electrical safety published every five years by the IEEE Standards Association, is now in a key stage of its 2028 update.
ANSI Invites Feedback on Proposed International Committee for Digital Product Passports
The American National Standards Institute (ANSI) is seeking comments by October 9 on a proposal to create a new international committee for Digital Product Passports (DPPs).
Food Safety ConnectED Portal Invites Professionals to Mentor Students
The Food Safety ConnectED portal is inviting food safety professionals to join as mentors, helping students and educators connect with experienced experts in the field.
ANSI Launches Compliance Platform Covering Standards and Regulations
The American National Standards Institute (ANSI) has launched Compliance Solutions,
Modernizing Accreditation: ANAB Launches Digital Technology Initiative and AI Task Force
The ANSI National Accreditation Board (ANAB) has launched its Digital Technology Initiative (DTI) to advance digital transformation across its accreditation services.
USNC Current Summer Edition Highlights Education in Standards
The new Summer 2025 edition of the USNC Current focuses on how education and training can strengthen the standards workforce and prepare future leaders.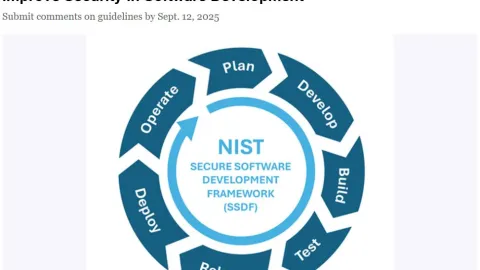
NIST Draft Guidelines Target Stronger Software Security
The U.S. National Institute of Standards and Technology (NIST) has released Special Publication 1800-44, a draft set of practices aimed at improving security throughout the software development process.
EPA’s WaterSense Program Honored With Ronald H. Brown Standards Leadership Award
The U.S. Environmental Protection Agency (EPA) WaterSense Program will receive the Ronald H. Brown Standards Leadership Award,
New HACCP Model Published to Guide Safe Production of Fermented, Salt-Cured, and Dried Foods
The U.S. Department of Agriculture (USDA) Food Safety and Inspection Service (FSIS) has released a new HACCP model designed to help meat and poultry establishments produce safe ready-to-eat shelf stable fermented, salt-cured, and dried products.
U.S. Standards Strategy 2025 Opens for Public Comment
The American National Standards Institute (ANSI) has released the draft of the 2025 United States Standards Strategy for public review.Global News

PEFC Opens Nominations For Forest Management Working Group Seats
The Programme for the Endorsement of Forest Certification (PEFC) is inviting nominations for its Sustainable Forest Management Working Group, with submissions open until April 22, 2026.
"Global Standard" Opens Second Consultation On Responsible Textile Standard GRTS
Global Standard has opened a second public consultation on Draft 2 of the Global Responsible Textile Standard (GRTS),
DIN SPEC 14027 Establishes First Comprehensive Framework for Corporate Security
The German Institute for Standardization (DIN) has published DIN SPEC 14027,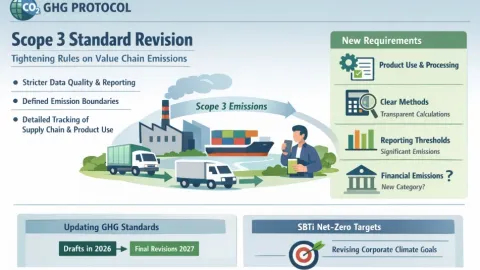
GHG Protocol Begins Scope 3 Revision Tightening Data and Measurement Rules
The Greenhouse Gas Protocol (GHG Protocol) has launched a major revision of its Scope 3 standard,
ISO 17020 Third Edition Published With Risk-Based Approach
A new edition of ISO/IEC 17020 has been published, introducing updated requirements for inspection bodies and replacing the 2012 version after a technical revision.
ISO TS 20125-1 for Eco-Design of Digital Services Released
The International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) have published ISO/IEC TS 20125-1:2026,
ISO 19650 Parts 1 and 2 on Information Management in BIM Projects Open for Consultation
The International Organization for Standardization (ISO) has opened a public consultation on proposed changes to ISO 19650 Parts 1 and 2,
TIA Develops First Data Center Infrastructure Quality Standard DCE 9000
The Telecommunications Industry Association (TIA) is developing DCE 9000, the first quality management system (QMS) standard specifically designed for modern data center physical infrastructure, with publication and certification planned for 2027.
ISO 18969 Clinical Evaluation Standard Draft Advances Past Voting Stage
Voting on the Draft International Standard (DIS) for ISO 18969, Clinical evaluation of medical devices, closed on March 10, 2026, marking its progression to stage 40.60 within the International Organization for Standardization (ISO).
GLOBALG.A.P. Replaces Technical Committees With Five Working Groups and Names Members
Agraya has replaced GLOBALG.A.P. technical committees with five technical working groups (TWGs) and appointed members to each group, with up to 16 experts per group across five areas.
Renault Group Updates Customer Requirements for IATF 16949 Suppliers
Renault Group has updated its Customer Specific Requirements (CSR) for IATF 16949:2016, with the new version taking effect in April 2026 and applying to all suppliers across its global operations.

