News United States of America
Please note that you have to be a registered member with paid membership in order to see full articles.
RegisterSelected News

National Working Group USA Expands Access to GLOBALG.A.P. Standards
The National Technical Working Group (NTWG) USA has made GLOBALG.A.P. standards more accessible to American producers by tailoring them to local laws and farming practices.
ANSI Seeks Proposals for Interactive Standards Exhibition at Innovation Summit
The American National Standards Institute (ANSI) is calling for proposals to take part in Standards Power Progress, an interactive exhibition at its inaugural Innovation Summit on October 21, 2025, in Rockville, Maryland.
ANSI Names Winners of 2025 Awards for Standards Leadership
The American National Standards Institute (ANSI) has announced the winners of its 2025 Leadership and Service Awards, recognizing 20 individuals for their impact on national and international standardization and certification efforts.
SGS Introduces Services to Support FSMA 204 Food Traceability Compliance
SGS has launched a set of services to help companies prepare for upcoming food traceability requirements in the United States.
FDA Shift to ISO 13485 Makes Live Quality Management System Essential for Medical Devices
Starting February 2, 2026, the U.S. Food and Drug Administration (FDA) will align its Quality System Regulation with ISO 13485:2016, making it the foundation for all medical device market submissions.
Nelson-Jameson Idaho Distribution Center Earns Perfect Score in First SQF Audit
Nelson-Jameson's distribution center in Jerome, Idaho, has earned a perfect 100% score in its first audit under the Safe Quality Food (SQF) Program, reflecting the highest level of food safety and quality certification.
ANSI Seeks Feedback on New ISO Proposal to Standardize Fundamentals of Services
The American National Standards Institute (ANSI) is requesting public comments by September 4, 2025, on a proposed new ISO technical committee aimed at developing global standards for the fundamentals of services.
Breakdown of Changes in the Revised ASTM E2659 Certificate Program Standard
ANSI/ASTM E2659-24, the 2024 edition of the Standard Practice for Certificate Programs, introduces significant updates to terminology, program structure, and certificate management.
How U.S. Standards Bodies Are Shaping the Leaders of Tomorrow
Several U.S. standards organizations are ramping up efforts to engage and prepare young professionals, students, and educators for careers in standardization and STEM-related fields.
ANSI Forms Technology Consortium Council to Boost Standards Collaboration
The American National Standards Institute (ANSI) has created a new Technology Consortium Council (TCC) to strengthen collaboration and dialogue between consortia and the broader standardization community.
GLOBALG.A.P. Compliance Guide Published for U.S. Fruit and Vegetable Farms
A national interpretation guideline has been published to help United States producers meet the requirements of the Integrated Farm Assurance (IFA) v6 GFS standard for fruit and vegetables,
NIST Standard Enables Accurate Dosage of New Anticancer Drugs
The National Institute of Standards and Technology (NIST) has developed the first U.S. standard for measuring the radioactivity of actinium-225, a key ingredient in a new class of anticancer drugs.Global News

ISO Releases Practical Guide To Integrating Multiple Management Systems
ISO has released a new publication, Integrated management systems – A practical guide (IMS PG), offering organizations a clear way to combine multiple management system standards into one coherent system.
ISO TR 25326 Gathers Global Examples Of Green Logistics In Practice
The International Organization for Standardization (ISO) has published ISO/TR 25326:2026, a new international standard that compiles case studies of green logistics to support lower-carbon operations across global supply chains.
BYD Joins IATF As First NEV-Only Automaker And Second Asian Member
The International Automotive Task Force (IATF) has approved the addition of BYD Group as a new member of IATF AISBL,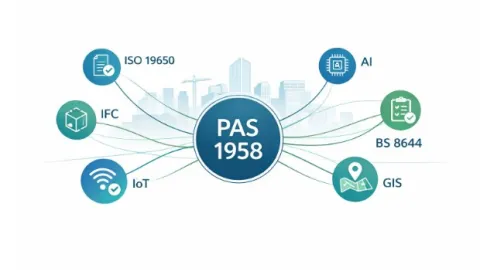
PAS 1958 Published To Connect Built Environment Data Standards
The British Standards Institution (BSI) has published PAS 1958:2026, a new guide that helps organizations understand how existing data and information standards in the built environment, including buildings and infrastructure, work together within a single framework.
International Quality Awards 2026 Finalists Announced
The Chartered Quality Institute (CQI), a professional body for quality management, has announced the finalists for its International Quality Awards 2026,
ASC Chain Of Custody Set To Become Supply Chain Module In Major Overhaul
The Aquaculture Stewardship Council (ASC), an international non-profit that sets standards for responsible seafood farming,
ISO 22000 Revision Enters Public Enquiry Stage
The International Organization for Standardization (ISO) has moved the revision of ISO 22000 to the public enquiry stage.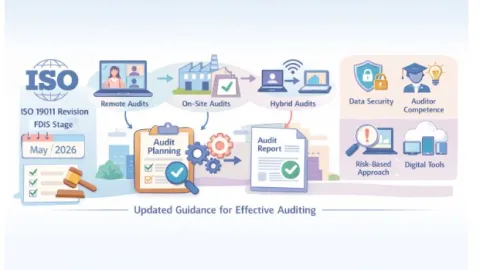
ISO 19011 Auditing Standard Enters Final Stage With Hybrid And Digital Shift
The International Organization for Standardization (ISO) has advanced the revision of ISO 19011 to the Final Draft International Standard (FDIS) stage.
IMDRF Playbook Guides Global Use Of Shared Medical Device Regulatory Reviews
The International Medical Device Regulators Forum (IMDRF) has published a playbook to help regulators use shared approaches when reviewing medical devices, known as regulatory reliance.
DIN Introduces First Framework For AI Testing Tools
The German Institute for Standardization (DIN) has published DIN SPEC 92006, introducing the first unified framework for artificial intelligence (AI) testing tools and setting clear requirements for how these tools should be designed and used.
FSSC 22000 Version 7 Release Moved To May 2026
The Foundation for Food Safety Certification (FSSC) has delayed the launch of FSSC 22000 Version 7, with publication now expected in early May 2026.

